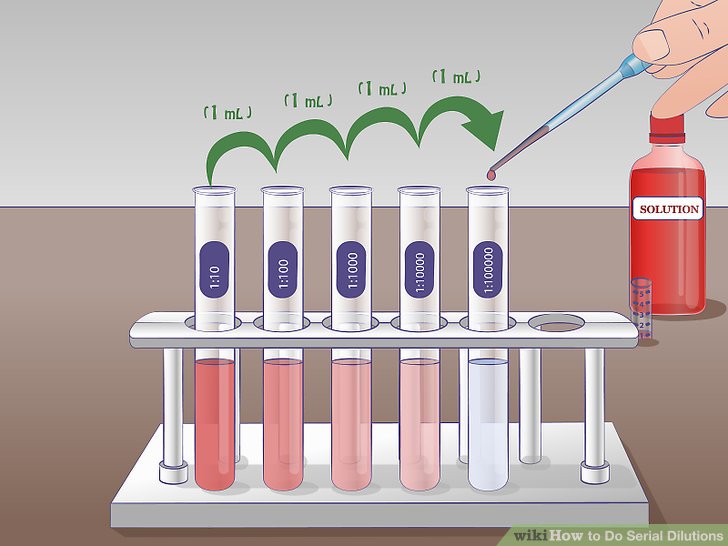
A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. A ten-fold serial dilution could be 1 M, 0.1 M, 0.01 M, 0.001 M .. Serial dilutions are used to accurately create highly diluted solutions as well as solutions for experiments resulting in concentration curves with a logarithmic scale. A tenfold dilution for each step is called a logarithmic dilution or log-dilution, a 3.16-fold (100.5-fold) dilution is called a half-logarithmic dilution or half-log dilution, and a 1.78-fold (100.25-fold) dilution is called a quarter-logarithmic dilution or quarter-log dilution. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
Serial Dilution Steps Calculator
In biology and medicine[edit]
A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion. Serial dilutions are much easier to make and they cover the range evenly. So, you need to make a 1/1,000 dilution to start with. Then you need to serially dilute that 2-fold per step in five steps. You could make 1/1,000 by adding 1 microliter of sample to 0.999 ml diluent. Dec 26, 2013 This feature is not available right now. Please try again later. The first step in making a serial dilution is to take a known volume (usually 1ml) of stock and place it into a known volume of distilled water (usually 9ml). This produces 10ml of the dilute solution. This dilute solution has 1ml of extract /10ml, producing a 10-fold dilution.
In biology and medicine, besides the more conventional uses described above, serial dilution may also be used to reduce the concentration of microscopic organisms or cells in a sample. As, for instance, the number and size of bacterial colonies that grow on an agar plate in a given time is concentration-dependent, and since many other diagnostic techniques involve physically counting the number of micro-organisms or cells on specials printed with grids (for comparing concentrations of two organisms or cell types in the sample) or wells of a given volume (for absolute concentrations), dilution can be useful for getting more manageable results.[1] Serial dilution is also a cheaper and simpler method for preparing cultures from a single cell than optical tweezers and micromanipulators.[2]
Steps Of Serial Dilution
In homeopathy[edit]
Serial dilution is one of the core foundational practices of homeopathy, with 'succussion', or shaking, occurring between each dilution. In homeopathy, serial dilutions (called potentisation) are often taken so far that by the time the last dilution is completed, no molecules of the original substance are likely to remain.[3][4]
See also[edit]

References[edit]
- ^K. R. Aneja. Experiments in Microbiology, Plant Pathology and Biotechnology. New Age Publishers, 2005, p. 69. ISBN81-224-1494-X
- ^Booth, C.; et al. (2006). Extremophiles. Methods in microbiology 35. Academic Press. p. 543. ISBN978-0-12-521536-7.
- ^Weissmann, Gerald (2006). 'Homeopathy: Holmes, Hogwarts, and the Prince of Wales'. The FASEB Journal. 20 (11): 1755–1758. doi:10.1096/fj.06-0901ufm. PMID16940145. Retrieved 2008-02-01.
- ^Ernst, Edzard (November 2005). 'Is homeopathy a clinically valuable approach?'. Trends in Pharmacological Sciences. 26 (11): 547–548. CiteSeerX10.1.1.385.5505. doi:10.1016/j.tips.2005.09.003. PMID16165225.
- Michael L. Bishop, Edward P. Fody, Larry E. Schoeff. Clinical Chemistry: Principles, Procedures, Correlations. Lippincott Williams & Wilkins, 2004, p. 24. ISBN0-7817-4611-6.
External links[edit]
- How to Make Simple Solutions and Dilutions, Bates College
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Serial_dilution&oldid=904833671'
Learning Objective
- Calculate the concentration of a diluted solution.
Key Points
- Most commonly, a solution’s concentration is expressed in terms of mass percent, mole fraction, molarity, molality, and normality. When calculating dilution factors, it is important that the units of volume and concentration remain consistent.
- Dilution calculations can be performed using the formula M1V1 = M2V2.
- A serial dilution is a series of stepwise dilutions, where the dilution factor is held constant at each step.
Terms
- dilutiona solution that has had additional solvent, such as water, added to make it less concentrated
- serial dilutionstepwise dilution of a substance in solution
Dilution refers to the process of adding additional solvent to a solution to decrease its concentration. This process keeps the amount of solute constant, but increases the total amount of solution, thereby decreasing its final concentration. Dilution can also be achieved by mixing a solution of higher concentration with an identical solution of lesser concentration. Diluting solutions is a necessary process in the laboratory, as stock solutions are often purchased and stored in very concentrated forms. For the solutions to be usable in the lab (for a titration, for instance), they must be accurately diluted to a known, lesser concentration.
Armored Core for Answer PS3 Full Version (USA + EUR) ISO & PKG, Download Armored Core for Answer Xbox 360 Jtag/RghRegion Free ISO + Fix Update DLC Free. Armored Core for Answer ps3 iso, Download game ps3 iso, hack game ps3 iso, dlc game save ps3, guides cheats mods game ps3, torrent game ps3. Armored Core 4 PS3 Game ISO, Download Armored Core 4 PS3 (USA) ISO & PKG, Armored Core 4 Game Full Version (USA - BLUS30027) ISO Download + Fix DLC Free. Some frequently asked question answers about Armored Core 4 game. Q: Can CFW and OFW, both users install Armored Core 4. Armored core 4 answer iso. Jan 19, 2018 The game released on September 16, 2008, for Xbox 360. Armored Core for Answer game is 3D vehicular combat Mecha game which is based on action. The game is the 13th installment in the series of Armored Core games. Armored Core For Answer Xbox 360 game is launched with a new interface and many parts are featured which is not found in the Armored.
The volume of solvent needed to prepare the desired concentration of a new, diluted solution can be calculated mathematically. The relationship is as follows:
[latex]M_1V_1=M_2V_2[/latex]
M1 denotes the concentration of the original solution, and V1 denotes the volume of the original solution; M2 represents the concentration of the diluted solution, and V2 represents the final volume of the diluted solution. When calculating dilution factors, it is important that the units for both volume and concentration are the same for both sides of the equation.
Example
- 175 mL of a 1.6 M aqueous solution of LiCl is diluted with water to a final volume of 1.0 L. What is the final concentration of the diluted solution?
- [latex]M_1V_1=M_2V_2[/latex]
- (1.6 M)(175 mL) = M2(1000 mL)
- M2 = 0.28 M
Serial Dilutions
Serial dilutions involve diluting a stock or standard solution multiple times in a row. Typically, the dilution factor remains constant for each dilution, resulting in an exponential decrease in concentration. For example, a ten-fold serial dilution could result in the following concentrations: 1 M, 0.1 M, 0.01 M, 0.001 M, and so on. As is evidenced in this example, the concentration is reduced by a factor of ten in each step. Serial dilutions are used to accurately create extremely diluted solutions, as well as solutions for experiments that require a concentration curve with an exponential or logarithmic scale. Serial dilutions are widely used in experimental sciences, including biochemistry, pharmacology, microbiology, and physics.
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
http://www.boundless.com/
Boundless Learning
CC BY-SA 3.0.
Boundless Learning
CC BY-SA 3.0.
http://en.wikipedia.org/wiki/serial%20dilution
Wikipedia
CC BY-SA 3.0.
Wikipedia
CC BY-SA 3.0.
http://en.wiktionary.org/wiki/dilution
Wiktionary
CC BY-SA 3.0.
Wiktionary
CC BY-SA 3.0.
http://en.wikipedia.org/wiki/Serial_dilution
Wikipedia
CC BY-SA 3.0.
Wikipedia
CC BY-SA 3.0.
“Sunil Kumar Singh, Dilution. September 17, 2013.â€
http://cnx.org/content/m17123/latest/
OpenStax CNX
CC BY 3.0.
OpenStax CNX
CC BY 3.0.
http://commons.wikimedia.org/wiki/File:Dilution-concentration_simple_example.jpg
Wikimedia
CC BY-SA.
Wikimedia
CC BY-SA.
 RSS Feed
RSS Feed
